Patients' High Acceptability of a Future Therapeutic HIV Vaccine in France: A French Paradox?
Réseau Ville Hôpital Val de Seine, Foch Hospital (Dimi, Zucman, Majerholc); University Paris-Diderot (Chassany, Lalanne, Aubin-Auger, Duracinsky); Fernand Widal Hospital (Chassany, Duracinsky); Regional Hospital Orléans (Prazuck); Louis Mourier Hospital (Mortier); Observatoire régional de la santé Paca (Verger); SESSTIM-IRD (Verger); Bicetre Hospital (Duracinsky)
The World Health Organization (WHO) Strategic Advisory Group of Experts (SAGE) on Immunization has recognised that reservations about vaccines are a growing global problem. Thus, anticipation of the acceptability of vaccines by the public has become an increasingly important factor in the development of new vaccines, such as an HIV therapeutic vaccine. In the French general population, refusal of vaccination has become frequent. In this context, the Representations and Acceptability of a Therapeutic HIV Vaccine (RAVVIH) study was designed to assess the acceptability of a future HIV therapeutic vaccine in HIV-positive outpatients aged 18-75 years in French AIDS-care centres.
Seventy percent (N=151) of those surveyed in early 2014 were men, and almost all patients (96%) were taking antiretroviral treatment (ART). The patients' medical files revealed that immunisation coverage for hepatitis B, diphtheria, tetanus and poliomyelitis was high, at 75%. Immunisation coverage was much lower for influenza (15%) and pneumococcus (21%).
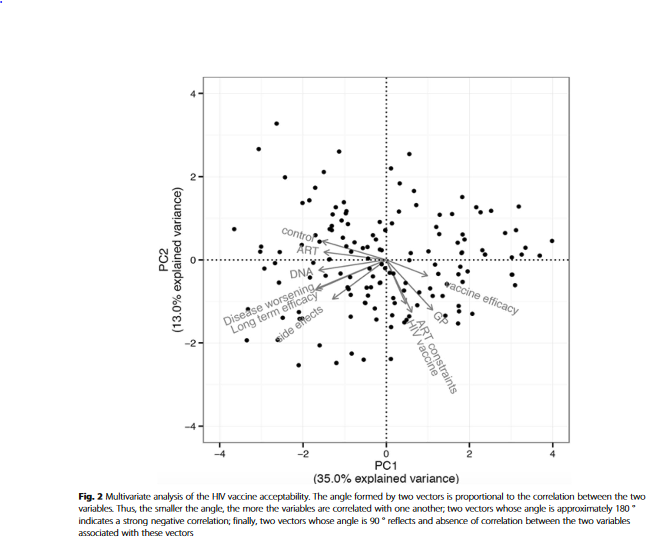
When interviewed about a possible future therapeutic HIV vaccine, the mean score for acceptability on the vaccine acceptability score (VAS) was 8.4 (out of 10), and 92% of patients agreed to be vaccinated if such a vaccine becomes available. Acceptability was strongly correlated with the confidence in the treating physician; even some patients who believed that doctors in general would not tell all the truth about vaccines would have a strong trust in their physician and would agree to their advice. HIV therapeutic acceptance depended on the expected characteristics of the vaccine, notably its duration of effectiveness: 44% of participants demanded the vaccine to be effective for life. The mode of administration (syringe) was not a barrier for these participants (86%), although 71% of participants feared possible side effects. Willingness to participate in a therapeutic HIV vaccine clinical trial was high (74%); only 9% of patients gave a negative response, and 17% had no opinion.
The researchers concede that it is possible that the 3 clinics participating in this study may be more actively encouraging updating preventive vaccination coverage than other hospitals. Also, the uptake of immunisation coverage of this study population was somewhat higher than the figures observed for other French hospitals. And it may be possible that in a real-life decision situation some patients would decide differently. All that said, the people living with HIV (PLWH) in this study were found to be receptive to the idea of receiving a therapeutic HIV vaccine.
BMC Infectious Diseases (2019) 19:401 https://doi.org/10.1186/s12879-019-4056-6
- Log in to post comments












































